 The Phase Change Matters e-mail newsletter is a weekly summary of the latest news and research on phase change materials and thermal energy storage. To subscribe, visit www.puretemp.com/subscribe. For more frequent updates, follow @puretemp on Twitter or visit the Phase Change Matters blog, www.puretemp.com/pcmatters.
The Phase Change Matters e-mail newsletter is a weekly summary of the latest news and research on phase change materials and thermal energy storage. To subscribe, visit www.puretemp.com/subscribe. For more frequent updates, follow @puretemp on Twitter or visit the Phase Change Matters blog, www.puretemp.com/pcmatters.
PHASE CHANGE MATERIAL
PCM-based free cooling in buildings: A way forward
A review of research on PCM-based free cooling in buildings suggests that increased thermal conductivity, governmental support and public awareness are essential to wider adoption of the technology. Among the other conclusions by a team of researchers at two universities in India:
• The free-cooling concept is best suited for less humid and maximum diurnal temperature range regions. However, with careful design of heat exchangers along with dehumidification of air, free cooling can be effectively implemented even in warm and humid areas.
• By decreasing the inlet air temperature with optimal inlet air velocity, solidification time can be reduced and thus the PCM can be charged in less time.
• Supercooling can be reduced by adding nucleating agents but cannot be mitigated completely.
• Mapping of free-cooling potential zones and construction of large-scale demonstration projects are essential to making the technology commercially viable.
• Commercializing and mass implementation of free-cooling technology in residential sectors will curtail air conditioner use and reduce greenhouse gas emissions.
Global PCM market projected to hit $1.67 billion by 2020
The global market for phase change material, which was valued at $563.5 million in 2014, is expected to reach $1.67 billion by 2020, according to a report by Mordor Intelligence. That’s a compound annual growth rate of 19.9 percent.
The report attributes some of the projected growth to demand for high-performance construction materials in emerging economies such as India, Brazil, China, Russia and South Africa.
Companies profiled in the report include BASF, Rubitherm Technologies, DuPont, Pluss Advanced Technologies, Phase Change Energy Solutions, China National Bluestar Group and Entropy Solutions.
India’s high-altitude shelters use PCM, water tanks to store thermal energy
India’s Defense Research and Development Organization (DRDO) has established the highest research station on Earth. The Chang La station, near Pangong Lake in northern India, stands at 17,500 feet above sea level. Along with the station, DRDO is also introducing a new technology for solar-heated shelters. According to the Tribune of India:
“DRDO has also developed a unique green energy based micro-climate-controlled shelter for use in high-altitude areas that traps and utilises solar energy to maintain the temperature inside. DRDO scientists said while power generated from solar energy could be stored in batteries, it is very difficult to store heat energy derived from the sun for later use. The new shelter, called Dhruv, uses phase change materials for night applications, heat trapping roof design, freeze-resistant underground water storage tanks and solar energy based thermal budgeting.”
Rare and endangered plants will be preserved at the Chang La station, where temperatures can fall as low as -40º C. The station will also be used to conduct extreme-altitude research on plants, animals and bio-medical material. The station will house up 20 scientists and support staff. Solar power will be the primary source of energy.
Material ConneXion adds PureTemp to NYC library, online database
PureTemp phase change materials have been accepted for display at Material ConneXion‘s flagship library in New York City. PureTemp, the world’s first 100 percent renewable PCM, will also be listed in Material ConneXion’s comprehensive online database, starting in November 2015.
Material ConneXion is the world’s largest membership-based library, with more than 7,500 unique, innovative and sustainable materials and seven locations worldwide. Each month, an independent jury selects materials and processes for inclusion in the library.
PATENT APPLICATIONS
Core of a capsule comprising phase change material
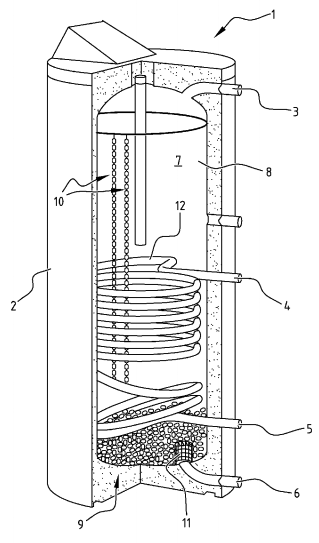 U.S. patent application 2015028556 (applicant Flamco BV, Netherlands):
U.S. patent application 2015028556 (applicant Flamco BV, Netherlands):
“The present invention relates to a core of a capsule, capsules having the cores comprising phase changing material (PCM) to be processed to be form retaining in particular during subsequent steps of applying a skin to form the capsule, as well as an assembly of a plurality of such capsules, use thereof in a buffer, and a production method for manufacturing such capsules or assemblies, for instance in strings. …
“The invention is based on the insight that the buffers in heating systems or heat exchange systems, like central heating systems, are usually filled with heat exchange fluids, like water. However, water and other usual heat exchange fluids have a limited heat capacity. According to the inventors of the present invention, it has been reported that phase change materials could be employed to enhance the heat capacity of a buffer, but to date no viable embodiment has been encountered.”
System for reversibly storing electrical energy as thermal energy
U.S. patent application 20150276326 (assignee Alstom Technology Ltd., Switzerland):
“The system can include a reversible subcritical vapour-liquid cycle energy storage system with a single hot storage fluid tank and cold storage fluid tank that are inter connected by an inter storage tank flow path. The inter storage tank flow path includes an inter storage heat exchanger in the vapour-liquid cycle that enables sensible heat transfer between the working fluid and storage fluid as the storage fluid passes between the hot storage fluid tank and the cold storage fluid tank. This supplements latent heat transfer between the working fluid and the hot storage fluid tank and the cold storage fluid tank.”
SOLAR ENERGY
NREL lays off 15 solar energy researchers, plans further cuts at Colorado lab
The National Renewable Energy Laboratory in Golden, Colo., laid off 15 solar energy researchers this week and announced plans to eliminate 50 to 60 other positions via buyouts. Reduced funding for the U.S. Department of Energy’s Solar Energy Technologies Office – from $289 million in 2012 to $233 million this year – forced NREL to shift the priorities of its solar research program, spokesman George Douglas told the Denver Post. Additional cuts will come in areas such as business systems, information technology and public affairs.
PACKAGING
The case for a new cold chain performance standard
Peli BioThermal design manager Richard Wood endorses the adoption of Kelvin hours (KH) as a new performance standard for the global temperature-controlled packaging industry:
“The benefits of adopting KH as a standard performance measure is that it does not tie itself to a particular ambient test profile. Rather, it is centred on what the specific selected shipping system is capable of doing; therefore, you can say that a particular shipping system will perform against ambient profile X, Y or Z for a specific number of hours for those different demands. It makes for a less subjective and more measured approach. KH could be collated via a simple equation: average thermal stress (K) x control duration (hrs) = Khrs. Adopting this calculated stress testing, using vendor qualification data, would enable simple, fit for purpose evaluations.“KH could easily be adopted as a tool to select the most appropriate level of performance of packaging. This could be used across the board, with a broad range of performance points for the newer-style phase change material (PCM) systems, which is where the KH measure works particularly well.”
NEW PRODUCTS

Creators of food-warming mat seek funding on IndieGoGo
The creators of a PCM-laden mat designed to keep food warm for up to an hour have launched an IndieGoGo campaign to raise funds for production and distribution.
The thermal mat, dubbed TheM, uses Outlast phase change technology to absorb and store heat from a hot dish, and then release it back to the dish as needed to keep food at 150º F. The octagonal 8-inch mat weighs less than 2 pounds and is less than a quarter-inch thick. It remains cool to the touch as it absorbs and releases heat.
ACADEMIC RESEARCH
Exception to rule may lead to better understanding of thermal expansion
 It’s widely known, at least among people who study such things, that scandium trifluoride is a thermal rule breaker. Most materials expand when they are heated and shrink when they are cooled. Scandium trifluoride does the opposite. University of Connecticut physicist Jason Hancock has been conducting research on the material that may lead to a better understanding of thermal expansion. UConn Today’s Kim Krieger explains:
It’s widely known, at least among people who study such things, that scandium trifluoride is a thermal rule breaker. Most materials expand when they are heated and shrink when they are cooled. Scandium trifluoride does the opposite. University of Connecticut physicist Jason Hancock has been conducting research on the material that may lead to a better understanding of thermal expansion. UConn Today’s Kim Krieger explains:
“Hancock and graduate student Sahan Handunkanda decided to look at scandium trifluoride because its odd behavior might give them some clues on what to look for in more typical materials. Not only does scandium trifluoride drastically shrink as it warms over a huge range of temperature (almost 1,100K or 2,000 F); it also keeps the same, stable cubic crystal structure over an even larger temperature range, from near absolute zero to 1,800 degrees Kelvin (2,780 degrees Fahrenheit), at which point it melts. Very few materials can boast of being so stable; most have some kind of phase change, during which their atoms shift positions, at least once when they’re warmed over 2,780 degrees Fahrenheit.”
RESEARCH ROUNDUP
For our full list of recent academic research, see puretemp.com/academic. Here are highlights from the past week:
From Procedia Engineering:• PCM-based High-density Thermal Storage Systems for Residential and Small Commercial Retrofit Applications
• Review of Phase Change Materials Integrated in Building Walls for Energy Saving
• Research on Indoor Thermal Environment Improvement of Lightweight Building Integrated with Phase Change Material under Different Climate Conditions
• Estimating the Adaptability of Phase Change Material Board on Building Envelope of Telecommunications Base Stations
• Simulation on Phase Change Thermal Storage Panel Based on Capillary Network
From Energy Conversion and Management:
• Experimental analysis of a low cost phase change material emulsion for its use as thermal storage system
• Form-stable LiNO3–NaNO3–KNO3–Ca(NO3)2/calcium silicate composite phase change material (PCM) for mid-low temperature thermal energy storage
From Energy and Buildings:
• Shape-stabilized phase change materials based on fatty acid eutectics/expanded graphite composites for thermal storage
From International Communications in Heat and Mass Transfer:
• Experimental study on melting/solidification and thermal conductivity enhancement of phase change material inside a sphere
From Journal of Technology Innovations in Renewable Energy:
• Numerical Prediction of Thermal Storage Using Phase Change Material
From Sustainable Chemistry and Engineering:
• Encapsulation of Phase Change Material with Water-Absorbable Shell for Thermal Energy Storage
From Chemie der Erde – Geochemistry:
• Great new insights from failed experiments, unanticipated results and embracing controversial observations
NETWORKING
Connect with PCM experts and industry leaders on LinkedIn
More than 370 of your peers have joined a new LinkedIn group devoted to the discussion of phase change material and thermal energy storage. The Phase Change Matters group is an interactive complement to the blog and newsletter of the same name.
You are invited to join the group and connect with PCM and TES experts from around the world. New members this week include Rok Lončarič, CEO at Cooling Inno; Tomer Rozenfeld, Ph.D. student at Ben Gurion University of the Negev; Gerard Oortman, director at PCM Technology and Salca BV; Deirdre Naehle, North American operations manager for va-Q-tec AG; and Dimitris Nikoleris, business development manager for Synectic Systems Group Ltd. Dimitris writes:
“I work in the marine industry. Have experience with heat pipes as means of heat superconductors for a number of different industries (electronics, WHR, solar). I have always been excited about PCM and the potential of storing heat. I am trying to find any documented applications of PCMs in the marine industry. Any input from the experts is very welcome.”
YOUR TURN
Got a question about PCMs or TES? Ask our experts
Two Entropy Solutions advisors, Dr. Mohammed Farid of the University of Auckland and Lucas B. Hyman of Goss Engineering, are ready to answer your questions about phase change material and thermal energy storage. We select the best questions sent to inquiries@puretemp.com and post the answers here each Friday.
