 The Phase Change Matters e-mail newsletter is a weekly summary of the latest news and research on phase change materials and thermal energy storage. To subscribe, visit www.puretemp.com/subscribe. For more frequent updates, follow @puretemp on Twitter or visit the Phase Change Matters blog, www.puretemp.com/pcmatters.
The Phase Change Matters e-mail newsletter is a weekly summary of the latest news and research on phase change materials and thermal energy storage. To subscribe, visit www.puretemp.com/subscribe. For more frequent updates, follow @puretemp on Twitter or visit the Phase Change Matters blog, www.puretemp.com/pcmatters.
PATENTS
System for energy consumption reduction and cost savings in a building
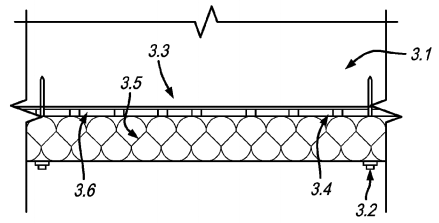
U.S. patent application 20170146251 (applicant Stasis Group Inc., Woodland, Calif.):
“A system for obtaining energy consumptions, savings and cost reduction in structures adapted for human habitation which includes the utilization of a plurality of mats including phase change material encapsulated within first and second layers of plastic material [3.6, above] having heat transfer capability disposed within the plenum area above a ceiling of a room within a building with the amount of phase change material contained within each mat being between 0.5 lbs. and 0.67 lbs. per square foot. … [The] solid to liquid transition point for said phase change material is from 72° F. to 76° F. and the liquid to solid transition point for said phase change material is from 71° F. to 68° F.”
Cordless tool system
U.S. patent application 20170149254 (applicant Chervon Ltd., Hong Kong):
“A tool discharges one battery pack and a charger simultaneously charges another battery pack to provide for continuous and circular operation of the tool. Tool system includes a tool and a lithium based battery pack having a nominal voltage of at least 56 volts for supplying power to the tool. A heat absorbing device is associated with the battery pack and includes a phase-change material. … [The] phase-change material comprises a mixture of sodium acetate, sodium dodecyl benzene sulfonate, carboxymethyl cellulose, and sodium carbonate.”
Related: Battery pack for energy storage devices (IntraMicron Inc., Auburn, Ala.)
Heat and humidity removal unit for a self-contained breathing apparatus
U.S. patent application 20170143933 (applicant Paragon Space Development Corp., Tucson, Ariz.):
“This disclosure relates to a regenerable, heat-abating, humidity-neutralizing, carbon dioxide removal system for a self-contained breathing apparatus. The self-contained breathing apparatus can include a carbon dioxide removal unit that scrubs carbon dioxide out of exhaled air from a user to provide humidified, scrubbed exhaled air. The self-contained breathing apparatus can further include a heat and humidity removal unit that is configured to receive the humidified, scrubbed exhaled air, and is configured to remove water vapor and heat associated with the water vapor from the humidified, scrubbed exhaled air in order to provide cooled, dehumidified inhalation air. The cooled, dehumidified air can be supplemented with oxygen and returned to the user at a comfortable temperature. In some implementations, the heat and humidity removal unit can replace conventional heat exchange and energy storage units, including heat exchange and energy storage units that use phase change materials.”
Tank for coolant liquid of a heat engine
U.S. patent application 20170145897 (applicant Hutchinson, Paris, France)
“A tank stores a coolant liquid and an engine and/or transmission lubricant for a motor vehicle heat engine, as well as a circuit for the cooling and heat regulation of that engine.
The tank has a thermally insulating outer wall including an inlet and an outlet for this liquid, and inside the wall, it comprises heating means that are able to preheat this liquid for the cold engine start of the vehicle. … According to one particularly advantageous embodiment of the invention, the tank comprises two other said compartments that are respectively made up of a first other compartment suitable for storing said engine lubricant and second other compartment suitable for storing said transmission lubricant, which each contain said at least one phase change material and which are separated from one another by another tight partition.”
Related: Exhaust heat regenerator for vehicle (Hanon Systems, South Korea)
Hybrid solar reactor and heat storage system
U.S. patent application 20170146262 (applicants University of Colorado, Denver; and Harper International Corp., Buffalo, N.Y.)
“A hybrid solar reactor and a heat storage system are disclosed. The hybrid solar reactor includes one or more heaters and a solar light guide assembly coupled to a shell of the reactor. The solar light guide assembly includes a solar light guide to direct solar energy to, for example, one or more reactor tubes within the shell. … Further embodiments of the disclosure relate to energy storage systems in which solar energy is used to heat a medium, such as phase change material (e.g., material that changes phase at or about 700° C.). The phase change material can be used to store heat that can be used when, for example, the energy storage system is off sun.”
IN BRIEF

• Roccor LLC of Longmont, Colo., is using phase change material in a device designed to manage internal heat in low-orbit satellites. “We have one product with paraffin wax inside a flat structure. When the spacecraft is hot, it dumps heat into that paraffin wax and turns that into a liquid — basically a store of energy,” Chris Pearson, Roccor’s vice president of space programs, said in an interview with Via Satellite. The heat is later released into the satellite to keep temperatures stable.
• A new research portal for energy-optimized construction summarizes some of the funding measures of the German Federal Ministry of Economics and Technology under one umbrella. The German-language site, www.projektinfos.energiewendebauen.de, also has research on thermal energy storage and provides examples of projects and activities that apply PCM to optimize the energy needs of buildings.
• Lloyds reports that Maersk Line is planning to make advanced monitoring data on premium perishables and pharma products available to its shipping customers later this year.
• New from Research and Markets: “Global District Cooling Market By Type and Region, Competition Forecast and Opportunities, 2012-2026“
• New from HJResearch: “Global Bio-Based Phase Change Materials Industry Market Research 2017“
• Pluss Advanced Technologies of Gurgaon, India, is seeking candidates for the position of research associate to design and perform experiments for the development of inorganic and organic phase change materials.
• The Swedish environmental group ChemSec has launched a free website linking companies selling alternatives to hazardous chemicals with those seeking to buy them. The first product request posted on The Marketplace: Clothing retailer H&M is looking for alternatives to the bisphenol A used in thermal paper for cash register and credit card receipts.
• Celsia Inc.‘s redesigned website, celsiainc.com, features a heat pipe performance calculator, a spreading delta-T calculator and a heat sink size calculator.
• Glacier Tek, maker of cooling vests powered by PureTemp phase change material, has launched a redesigned website at www.glaciertek.com. The new site features improved navigation, fresh photography and a new tagline: “Prepare to be Cool.”
• The City Council of Goderich, Ontario, has approved a proposal by NRStor to build a 1.75-megawatt compressed air energy storage plant in an unused salt cavern. The electricity created will feed into Ontario’s energy grid.
• The Expert Forum on VDI Guideline 2164, scheduled for June 20 in Düsseldorf, Germany, has been canceled. The forum, organized by the Association of German Engineers‘ building and building technology group in cooperation with the RAL Quality Association PCM, may be held at a later date.
RESEARCH ROUNDUP
For our full list of recent academic research, see puretemp.com/academic. Here are highlights from the past week:
From Renewable Energy:• Shape-stabilized composite phase change materials with high thermal conductivity based on stearic acid and modified expanded vermiculite
From AIMS Energy:
• Feasibility study of D-mannitol as phase change material for thermal storage
From Thermal Science and Engineering Progress:
• Computer simulations of heat transfer in a building integrated heat storage unit made of PCM composite
From European Polymer Journal:
• Preparation of acrylic based microcapsules using different reaction conditions for thermo-regulating textiles production
From Energy Procedia:
• Design of Experimental Investigation about the Effects of Flow Rate and PCM Placement on Thermal Accumulation
From Solar Energy Materials and Solar Cells:
• A eutectic salt high temperature phase change material: Thermal stability and corrosion of SS316 with respect to thermal cycling
• Novel form-stable phase change material composite for high-efficiency room temperature control
From Applied Thermal Engineering:
• Thermal Energy Storage System Operating with Phase Change Materials for Solar Water Heating Applications: DOE Modelling
From Procedia Engineering:
• A Comparative Study on the Effectiveness of Passive and Free Cooling Application Methods of Phase Change Materials for Energy Efficient Retrofitting in Residential Buildings
• Thermal Energy Storage Enhancement of Lightweight Cement Mortars with the Application of Phase Change Materials
From Renewable and Sustainable Energy Reviews:
• Thermal energy storage systems for concentrated solar power plants
From Construction and Building Materials:
• The effect of low temperature phase change material of hydrated salt on the performance of magnesium phosphate cement
NETWORKING
Connect with PCM experts and industry leaders on LinkedIn
More than a thousand of your peers have joined a LinkedIn group devoted to the discussion of phase change material and thermal energy storage. The Phase Change Matters group is an interactive complement to the award-winning blog and newsletter of the same name.
 You are invited to join the group and connect with PCM and TES experts from around the world. New members this week include Dr. Abdelhakim Hassabou, director of CPI International Group‘s International Energy Unit, Munich, Germany; Gary Hutchinson, president of Modality Solutions LLC, Los Angeles; Uver Villalobos, research assistant at Masdar Institute of Science and Technology, Abu Dhabi, United Arab Emirates; and Davinder Kumar, general manager R&D at TVS Motor Co., Krishnagiri, India.
You are invited to join the group and connect with PCM and TES experts from around the world. New members this week include Dr. Abdelhakim Hassabou, director of CPI International Group‘s International Energy Unit, Munich, Germany; Gary Hutchinson, president of Modality Solutions LLC, Los Angeles; Uver Villalobos, research assistant at Masdar Institute of Science and Technology, Abu Dhabi, United Arab Emirates; and Davinder Kumar, general manager R&D at TVS Motor Co., Krishnagiri, India.
Mohammad Rezvanpour, who is pursuing a master’s degree in engineering at the University of Tehran, joined the group earlier this year. He writes:
“Hello, my friends. I want to prepare nanocapsules with eicosane as a core and PMMA as a shell. I am following one paper to reduce the errors. But in this article (http://www.sciencedirect.com/science/article/pii/S0038092X12000199), in which the authors mention ‘dialysis tubing cellulose membrane,’ there is no further information about the exact characterization of this membrane and it is so important to know it. Unfortunately, the authors didn’t respond to my emails and I had to post my question here. Do you know what type of ‘Dialysis tubing cellulose membrane’ is the best for preparing the nanocapsules with above-mentioned circumstances?”